United States Endoscopy Devices Market Size and Forecast 2026–2034
Rising Demand for Minimally Invasive Diagnostics and Advanced Imaging Technologies Accelerates Market Expansion
Introduction
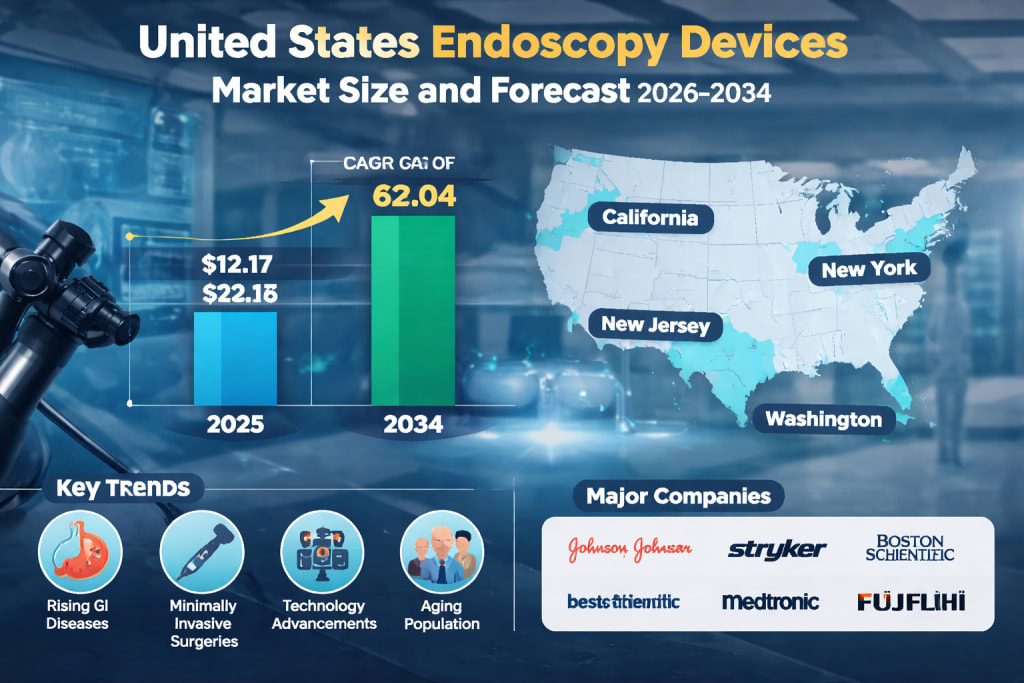
The United States Endoscopy Devices Market is poised for significant growth over the coming decade, driven by advancements in medical technology, the increasing prevalence of chronic diseases, and a rising preference for minimally invasive procedures. According to Renub Research, the market is projected to grow from US$ 12.17 Billion in 2025 to US$ 22.04 Billion by 2034, expanding at a Compound Annual Growth Rate (CAGR) of 6.82% between 2026 and 2034.
Endoscopy has become a cornerstone of modern diagnostic and therapeutic medicine. From gastrointestinal evaluations to respiratory and gynecological procedures, endoscopic technology enables physicians to examine internal organs with high precision while minimizing patient trauma. In the United States, the integration of advanced imaging, artificial intelligence, and robotics into endoscopic devices is transforming clinical practices and improving patient outcomes.
With the country's aging population, growing incidence of gastrointestinal disorders, and increasing adoption of outpatient surgical services, the demand for advanced endoscopy equipment is expected to rise steadily throughout the forecast period.
United States Endoscopy Devices Market Overview
Endoscopy devices are specialized medical instruments designed to visualize and treat internal organs without the need for large surgical incisions. These systems typically consist of an endoscope, a thin tube equipped with a camera, light source, and sometimes surgical tools that allow physicians to observe and operate inside the body.
Endoscopes can be flexible or rigid, depending on the medical application. They are commonly inserted through natural body openings such as the mouth, nose, or rectum, or through small surgical incisions. Physicians rely on these devices for diagnostic and therapeutic procedures across multiple medical specialties.
Examples of commonly used endoscopic devices include:
Gastrointestinal endoscopes
Bronchoscopes
Colonoscopes
Laparoscopes
In the United States, the demand for these devices is growing due to several factors, including the increasing incidence of gastrointestinal diseases, cancer, and respiratory disorders. Endoscopic procedures allow physicians to diagnose illnesses early, collect biopsy samples, and perform minimally invasive treatments.
Additionally, the country's well-established healthcare infrastructure, combined with increasing awareness of preventive health screening programs, has led to the widespread adoption of endoscopy technologies. Colonoscopy screening for colorectal cancer, for example, has become a routine preventive measure for millions of Americans.
The rise of ambulatory surgical centers (ASCs) and outpatient treatment facilities has also accelerated the demand for compact, efficient, and technologically advanced endoscopy equipment.
Growth Drivers of the United States Endoscopy Devices Market
Rising Prevalence of Gastrointestinal Diseases and Cancer
The growing burden of gastrointestinal (GI) diseases and cancer in the United States is a major factor driving the demand for endoscopy devices. These conditions often require precise diagnosis and monitoring through endoscopic procedures.
Statistics highlight the scale of the issue. In 2021, gastrointestinal healthcare expenditures reached approximately $111.8 billion. GI-related diagnoses or symptoms accounted for 14.5 million emergency department visits and 2.9 million hospital admissions across the country.
Furthermore:
315,065 new GI cancer cases were diagnosed
GI diseases contributed to 281,413 deaths
Around 23.5 million GI endoscopies were performed in 2022
In addition, the National Institutes of Health (NIH) allocated approximately $3.6 billion for gastrointestinal research in 2023, representing 7.4% of its total research budget.
Endoscopic procedures play a crucial role in diagnosing diseases such as:
Colorectal cancer
Gastroesophageal reflux disease (GERD)
Inflammatory bowel disease
Peptic ulcers
Early detection through colonoscopy and other endoscopic screenings improves survival rates and treatment outcomes. Preventive screening programs and national healthcare recommendations have further increased the adoption of endoscopic procedures.
According to projections, 2,041,910 new cancer cases and 618,120 cancer deaths are expected in the United States in 2025, reinforcing the need for advanced diagnostic tools such as endoscopy devices.
Growing Demand for Minimally Invasive Procedures
Another major driver of the market is the rising preference for minimally invasive surgical techniques. Endoscopic procedures provide several advantages compared to traditional open surgeries.
Key benefits include:
Smaller incisions
Reduced pain and trauma
Faster recovery times
Shorter hospital stays
Lower risk of complications
These benefits significantly improve patient experience while reducing healthcare costs.
Endoscopy is widely used for procedures such as:
Polyp removal
Tumor ablation
Internal bleeding control
Tissue biopsy
The rapid expansion of ambulatory surgery centers in the United States has further increased the demand for minimally invasive procedures. These facilities allow patients to receive treatment and return home the same day, improving convenience and reducing healthcare system strain.
Technological advancements such as high-definition imaging, flexible endoscopes, and robotic-assisted systems have further improved the accuracy and safety of these procedures.
For instance, in July 2025, ophthalmic device manufacturer BVI performed the first clinical procedure using its FDA-approved Leos™ Laser Endoscopy Ophthalmic System. The procedure was conducted by glaucoma specialist Dr. Nathan Radcliffe at the Ambulatory Surgery Center of Greater New York in the Bronx.
This milestone demonstrates the expanding role of endoscopic technology across various medical specialties.
Technological Advancements and Innovation in Endoscopy Devices
Rapid technological innovation is reshaping the endoscopy devices industry in the United States. Modern endoscopic systems are equipped with advanced imaging technologies such as high-definition (HD) and 4K visualization, improving diagnostic accuracy.
Emerging innovations include:
Capsule endoscopy
Robotic-assisted endoscopy
Artificial intelligence integration
Wireless imaging systems
These technologies allow physicians to detect abnormalities at earlier stages and perform complex procedures with greater precision.
For example, in January 2025, AnX Robotica received FDA clearance for the NaviCam capsule endoscopy system, enabling wireless diagnostic imaging of the small intestine.
Digital imaging systems and integrated software platforms also allow physicians to store and analyze endoscopic images, improving documentation and clinical decision-making.
As hospitals and surgical centers continue upgrading their medical infrastructure, demand for technologically advanced endoscopy devices is expected to rise significantly.
Challenges in the United States Endoscopy Devices Market
High Cost of Equipment and Procedures
Despite strong growth potential, the endoscopy devices market faces several challenges. One of the primary obstacles is the high cost of endoscopy equipment and procedures.
Advanced endoscopic systems require expensive components such as:
Video processors
High-definition imaging equipment
Flexible endoscopes
Surgical instruments
Healthcare facilities must also invest in ongoing maintenance, upgrades, and staff training. Smaller hospitals and clinics may struggle to afford these technologies.
For patients, endoscopic procedures can also be expensive, particularly for individuals with limited insurance coverage. Although many procedures are partially reimbursed, out-of-pocket costs remain a barrier for some patients.
Developing cost-effective technologies and affordable endoscopic solutions will be crucial for expanding access and sustaining market growth.
Infection Risks and Regulatory Compliance
Another challenge involves the risk of infection associated with improperly sterilized endoscopic devices. Reusable endoscopes require thorough cleaning and sterilization after every procedure to prevent cross-contamination.
Failure to maintain proper hygiene standards can result in healthcare-associated infections (HAIs), which may undermine patient trust and increase legal liabilities for healthcare institutions.
To address this issue, hospitals must invest in advanced sterilization equipment and implement strict infection-control protocols.
Additionally, endoscopy devices must undergo rigorous regulatory approval processes before entering the market. Manufacturers must comply with strict safety and quality standards, which can increase development costs and delay product launches.
While these regulations ensure patient safety, they can also create barriers for new entrants and increase operational expenses for device manufacturers.
United States Mechanical Endoscopic Equipment Devices Market
Mechanical endoscopic equipment devices play an essential role in therapeutic endoscopy procedures. These instruments assist physicians in performing specialized tasks during diagnostic or surgical interventions.
Examples of mechanical endoscopic devices include:
Forceps
Scissors
Snares
Dilators
Retrieval baskets
These tools are widely used for procedures such as tissue biopsy, polyp removal, foreign body retrieval, and treatment of gastrointestinal or respiratory diseases.
In the United States, the growing prevalence of digestive disorders and cancer is driving demand for these instruments. Mechanical devices enhance procedural precision and allow physicians to perform treatments without invasive surgery.
Technological improvements have also enhanced the durability, flexibility, and safety of these instruments, making them more reliable in clinical practice.
United States Bronchoscopy Devices Market
Bronchoscopy devices are used to diagnose and treat conditions affecting the lungs and airways. These instruments allow physicians to examine the bronchial tubes and collect tissue samples for laboratory testing.
Bronchoscopy procedures are commonly used to diagnose:
Lung cancer
Lung infections
Chronic obstructive pulmonary disease (COPD)
Airway obstructions
The rising incidence of respiratory diseases in the United States is increasing the demand for bronchoscopy procedures.
Modern bronchoscopy devices incorporate flexible bronchoscopes, high-definition imaging systems, and navigation technologies, which improve diagnostic accuracy and treatment efficiency.
As awareness of early lung cancer detection grows, hospitals and respiratory care centers are increasingly adopting advanced bronchoscopy technologies.
United States Laparoscopy Devices Market
Laparoscopy devices are widely used for minimally invasive surgical procedures involving the abdominal and pelvic cavities.
Common laparoscopic procedures include:
Gallbladder removal
Hernia repair
Gynecological surgeries
Laparoscopic surgery offers numerous advantages over traditional open surgery, including reduced pain, faster recovery, and smaller scars.
Technological advancements such as robotic-assisted laparoscopy and high-definition imaging systems have further improved surgical precision and patient safety.
The increasing number of surgical procedures and the rising prevalence of chronic diseases are contributing to the growing adoption of laparoscopy devices across U.S. healthcare facilities.
United States Endoscopy Devices Hospitals Market
Hospitals represent the largest end-user segment for endoscopy devices in the United States. A significant number of diagnostic and therapeutic procedures are performed in hospital settings.
Hospitals invest heavily in advanced medical technologies, including:
Flexible endoscopes
High-definition imaging systems
Specialized endoscopic instruments
These technologies improve diagnostic accuracy and support complex medical procedures.
Government healthcare initiatives, increasing healthcare expenditure, and growing awareness about early disease detection are also contributing to the rising demand for endoscopy devices in hospitals.
Key Regional Markets
California
California is one of the leading markets for endoscopy devices due to its advanced healthcare infrastructure and large population. The state is home to many prominent hospitals, research institutions, and medical device companies.
The increasing prevalence of chronic diseases such as colorectal cancer and gastrointestinal disorders is driving demand for endoscopic procedures. Additionally, California’s strong culture of medical innovation encourages the adoption of cutting-edge technologies like robotic-assisted endoscopy.
New York
New York is another major market for endoscopy devices due to its large aging population and high healthcare demand.
Healthcare providers in the state are actively adopting advanced endoscopic technologies to improve diagnostic capabilities and patient outcomes.
In March 2024, Scivita Medical Technology Co., Ltd. signed a strategic cooperation agreement with Boston Scientific Corporation to expand the development and distribution of endoscopic imaging systems and single-use endoscopes.
New Jersey
New Jersey’s strong pharmaceutical and medical device industry supports the growth of the endoscopy devices market in the state.
Hospitals and ambulatory surgery centers are increasingly adopting advanced endoscopic technologies to improve treatment efficiency and patient recovery times.
In March 2024, NTT Corporation partnered with Olympus Corporation to launch a pilot program for a cloud-based endoscopy solution that enables advanced image processing.
Washington
Washington has emerged as a growing hub for healthcare innovation and medical technology development.
Hospitals and research centers in the state are adopting next-generation endoscopy systems to improve diagnostic accuracy.
In May 2025, Olympus received FDA 510(k) clearance for its EZ1500 endoscopes featuring Extended Depth of Field technology, enhancing imaging clarity during procedures.
Competitive Landscape
The United States endoscopy devices market is highly competitive, with several global medical technology companies leading innovation and product development.
Major companies operating in the market include:
Johnson & Johnson
Stryker
Boston Scientific
CONMED
Medtronic Plc
Fujifilm Holdings
Smith and Nephew
These companies focus on product innovation, strategic partnerships, regulatory approvals, and acquisitions to strengthen their market presence.
Each company is analyzed across multiple parameters including company overview, key personnel, recent developments, SWOT analysis, and revenue performance.
Final Thoughts
The United States Endoscopy Devices Market is entering a period of sustained growth driven by technological innovation, increasing chronic disease prevalence, and the growing adoption of minimally invasive medical procedures.
As healthcare providers continue prioritizing early diagnosis and patient-centered care, endoscopic technologies will play an increasingly vital role in modern medicine. Innovations such as AI-assisted diagnostics, capsule endoscopy, and robotic systems are expected to further transform the landscape of diagnostic and surgical procedures.
With strong investments in healthcare infrastructure, research, and medical technology, the United States is well-positioned to remain a global leader in endoscopy innovation. Over the forecast period from 2026 to 2034, the market is expected to expand steadily, offering significant opportunities for device manufacturers, healthcare providers, and technology developers.




Comments
There are no comments for this story
Be the first to respond and start the conversation.